A strong acid will react with a strong base to form a neutral (pH = 7) solution.
What happens when you mix a strong acid with a strong base?
When a strong acid and a strong base are combined in the proper amounts—when [H+] equals [OH−]\)—a neutral solution results in which pH = 7. The acid and base have neutralized each other, and the acidic and basic properties are no longer present. Salt solutions do not always have a pH of 7, however.
What happens when acid mixed with base?
When an acid is added to a base, it will lead to the formation of salt and water molecules. This process of reaction of an acid and a base is known as the neutralization reaction.
What happens when strong acid reacts with weak base?
In a weak base-strong acid titration, the acid and base will react to form an acidic solution. … This results in a solution with a pH lower than 7. An example of this is the titration of hydrochloric acid (strong acid) into ammonia (weak base), which forms the conjugate acid ammonium and produces an acidic solution.Which type of reaction happens when a base is mixed with an acid?
The reaction of an acid with a base is called a neutralization reaction. The products of this reaction are a salt and water.
Why do reactions of mixing strong acid and strong base differ from the reaction of mixing weak acid and weak base?
Strong acids and strong bases refer to species that completely dissociate to form ions in solution. By contrast, weak acids and bases ionize only partially, and the ionization reaction is reversible. Thus, weak acid and base solutions contain multiple charged and uncharged species in dynamic equilibrium.
What does the complete reaction of a strong base and a strong acid produce what is this type of reaction called?
Neutralization Reactions When a strong acid and a strong base are combined in the proper amounts – when [H+] equals [OH−]\) – a neutral solution results in which pH = 7. The acid and base have neutralized each other, and the acidic and basic properties are no longer present.
When an acid reacts with a strong base which product always forms?
Well, whenever an acid and a hydroxide base are reacted together they always produce the same two types of products: a salt and water.Can a strong base neutralize a weak acid?
Weak Acid-Weak Base Neutralization When a strong base neutralizes a weak acid, the resulting solution’s pH will be greater than 7.
What happens if you touch a strong base?Bases with a pH greater than 10 can cause chemical burns. Strong bases include, calcium hydroxide, sodium hydroxide and potassium hydroxide. … Bases also react with oils on skin and fatty tissue, which can lead to extensive damage to skin and subcutaneous tissue.
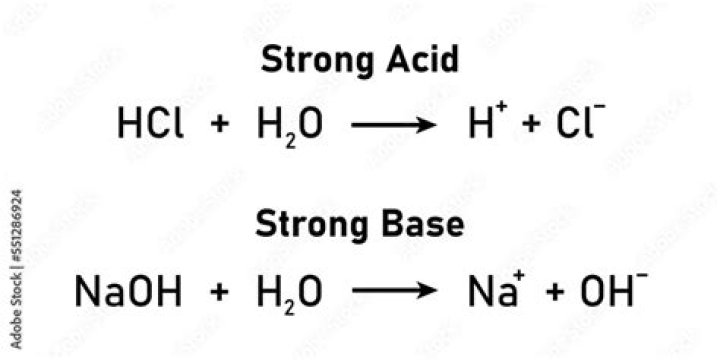
Article first time published onWhat happens when I mix a strong acid HCl with a strong base NaOH?
For example, if you mix hydrochloric acid (HCl) with sodium hydroxide (NaOH), the products formed are water (H20) and sodium chloride(NaCl), which is well-known as table salt.
When a strong acid combines with a strong base the products are a salt and water?
In such a neutralization reaction, the acid and the base will neutralize each other completely, thus resulting in a solution with a neutral pH. In fact, when a strong acid reacts with a strong base, the resulting products are water and an ionic salt.
What happens when a base reacts with a base?
When an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. The H(+) cation of the acid combines with the OH(-) anion of the base to form water. The compound formed by the cation of the base and the anion of the acid is called a salt.
What is a strong acid and a strong base?
Acids and bases that are completely ionized when dissolved in water are called strong acids and strong bases There are only a few strong acids and bases, and everyone should know their names and properties. These acids are often used in industry and everyday life.
When a strong acid is reacted with a strong base What is the net ionic equation?
So with a strong acid and strong base, the net ion equation is always H+ plus OH- yields H2O.
What is the difference between strong acid and weak acid give two examples of each?
Strong acid is an acid that ionizes completely in aqueous solution. … Weak acid is an acid that ionizes partially in a solution. It gives off only a few of its (H+) atoms when dissolved in water.
What will the acid base neutralization reaction between a strong acid plus a weak base yield?
Neutralization is the reaction between an acid and a base, producing a salt and a neutralized base. A strong acid yields a weak conjugate base (A–), so a strong acid is also described as an acid whose conjugate base is a much weaker base than water.
How does a strong acid differ from a weak acid?
the difference between a strong acid and a weak acid is in a strong acid,most of the molecules break up into ions in a solution. A weak acid has fewer molecules and they break up into ions. … the solution with a pH of 3 has a greater concentration of hydrogen ions.
What is the enthalpy of neutralization of strong acid and strong base?
the enthalpy of neutralisation of a strong acid and a strong base is always constant. the enthalpy of neutralization of a strong acid and a strong base is always considered constant because the strong acids and bases completely gets ionized in dilute solution. enthalpy of neutralization is always negative.
How are acids and bases harmful?
Acids and bases are corrosive substances that can attack various surfaces including human tissue. The amount of tissue damage they cause depends on the strength and concentration of the acid or base and the duration of exposure. The acidity or alkalinity of a substance can be determined by its pH value.
Why are base burns worse than acid burns?
Overall, alkaline agents are more toxic than acidic agents, due to the irreversible changes in protein and tissue damage.
Can a strong base burn you?
A very strong base has a pH of 14 and may also cause a severe burn. A substance with a pH of 7 is considered neutral and does not burn.
What happens when a strong acid such as HCl is added to the carbonic acid buffer system?
Bicarbonate-Carbonic Acid Buffer When sodium bicarbonate (NaHCO3), comes into contact with a strong acid, such as HCl, carbonic acid (H2CO3), which is a weak acid, and NaCl are formed. When carbonic acid comes into contact with a strong base, such as NaOH, bicarbonate and water are formed.
What happens when acid reacts with base temperature?
In the neutralization, acid and base react with each other and give salt and water as products. … Due to the production of heat, known as Heat of neutralization, the temperature of the solution increases. Hence, the correct answer is Option $(d)$.
What happens when an acid reacts with a base class 7?
Answer: When an acid reacts with a base, it neutralizes the base and forms salt and water as the products. The reaction involved is known as neutralization reaction.