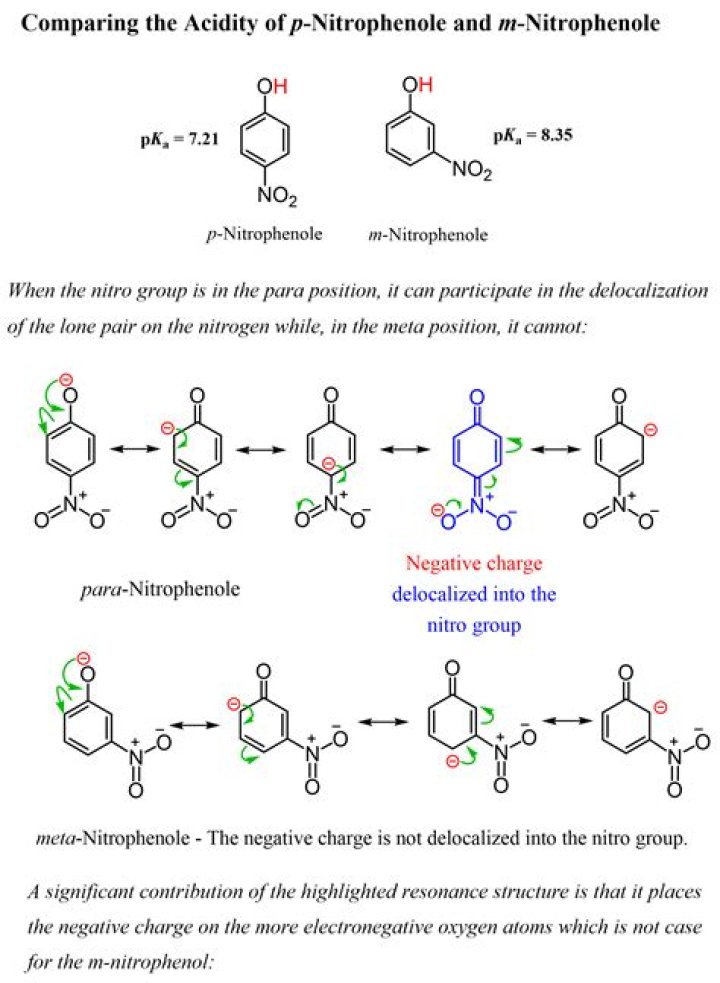
Red color is present in the visible spectrum and has a longer wavelength compared to other colors, this phenomenon is called the red shift. In an alkaline medium, p-nitrophenol shows red shift since the negatively charged oxygen molecules delocalise effectively compared to the unshared electron pair.
Why does p-nitrophenol shows red shift in alkaline medium?
Red color is present in the visible spectrum and has a longer wavelength compared to other colors, this phenomenon is called the red shift. In an alkaline medium, p-nitrophenol shows red shift since the negatively charged oxygen molecules delocalise effectively compared to the unshared electron pair.
What do you mean by Bathochromic shift?
Bathochromic shift (from Greek βαθύς bathys, “deep”; and χρῶμα chrōma, “color”; hence less common alternate spelling “bathychromic”) is a change of spectral band position in the absorption, reflectance, transmittance, or emission spectrum of a molecule to a longer wavelength (lower frequency).
What is P-nitrophenol used for in spectrophotometry?
The acyl esters of p-nitrophenol are the most commonly used model substrates to study the esterolytic activity of hydrolases. A quantitative detection of the released p-nitrophenolate can be easily carried out at 405–410 nm by spectrophotometry (Schmidt & Bornscheuer, 2005).What is blue shift in chemistry?
A blueshift is any decrease in wavelength (increase in energy), with a corresponding increase in frequency, of an electromagnetic wave. In visible light, this shifts a color towards the blue end of the spectrum.
Why P nitro phenol is more acidic than phenol?
The decrease in electron density of the O – H bond of p-nitrophenol, the polarity of O – H bond decreases. The electron withdrawing group (-NO ), withdraws electrons and disperses the negative charge. Therefore, -NO group stabilizes the phenoxide ion. Hence p-nitrophenol is more acidic than phenol.
Why does aniline show Hypsochromic shift in acid solution?
Answer: Aniline shows blue shift in acidic medium, it loses conjugation. When absorption intensity (ε) of a compound is increased, it is known as hyperchromic shift. If auxochrome introduces to the compound, the intensity of absorption increases.
How does NaOH stop enzyme reaction?
The NaOH has two effects: (l) it converts p-nitrophenol to p-nitrophenolate and develops the yellow color, and (2) it stops enzyme activity. By measuring the quantity of -nitrophenolate we are in effect measuring the quantity of -nitrophenol that was present (the product) before the addition of NaOH.Why does p-nitrophenol turn yellow?
pH indicator The yellow color of the 4-nitrophenolate form (or 4-nitrophenoxide) is due to a maximum of absorbance at 405 nm (ε = 18.3 to 18.4 mM−1 cm−1 in strong alkali).
What is bathochromic shift or red shift?BATHOCHROMIC SHIFT. The shift of absorption to a longer wavelength due to substitution or solvent effect (a red shift). … The shift of absorption to a shorter wavelength due to substitution or solvent effect (a blue shift).
Article first time published onWhat is red shift in chemistry?
Bathochromic shift: In spectroscopy, the position shift of a peak or signal to longer wavelength (lower energy). Also called a red shift. A hypsochromic shift is the shift of a peak or signal to shorter wavelength (higher energy).
What causes Hyperchromic shift?
An increase in the absorbtion of ultraviolet light by a solution of DNA as these molecules are subjected to heat, alkaline conditions, etc. The shift is caused by the disruption of the hydrogen bonds of each DNA duplex to yield single-stranded structures.
How does red Shift show that the universe is expanding?
Astronomers have discovered that, in general, the further away a galaxy is, the more red-shifted its light is. This means that the further away the galaxies are, the faster they are moving. … Red-shift data provides evidence that the Universe, including space itself, is expanding.
How do you calculate red Shift?
The redshift, symbolized by z, is defined as: 1 + z = l observed / l rest. z = 0.1. Note that if the observed wavelength were less than the rest wavelength, the value of z would be negative – that would tell us that we have a blueshift, and the galaxy is approaching us.
What does red shifting tell us about other galaxies and the universe?
Bottom line: A redshift reveals how an object in space (star/planet/galaxy) is moving compared to us. It lets astronomers measure a distance for the most distant (and therefore oldest) objects in our universe.
Why do we use UV spectroscopy?
UV/Vis spectroscopy is routinely used in analytical chemistry for the quantitative determination of different analytes, such as transition metal ions, highly conjugated organic compounds, and certain biological macromolecules. Measurement is usually carried out in solution.
What is UV visible spectroscopy discuss its principle?
The Principle of UV-Visible Spectroscopy is based on the absorption of ultraviolet light or visible light by chemical compounds, which results in the production of distinct spectra. Spectroscopy is based on the interaction between light and matter.
Why aniline behaves like that of benzene in acidic medium?
In case of aniline, the lone pair of electrons on nitrogen is available for donation and it is a less electronegative element than oxygen. Hence it will show basic character. Therefore, we can say that, in an acidic medium out of Aniline, Phenol, and Nitrobenzene, Aniline is the strongest Base.
Why P methyl phenol is less acidic than phenol?
Resonance in phenol: … Phenol is less acidic as compared to the above two because no electron withdrawing group is present and there is no inductive or resonance effect. Methyl phenol shows the electron donating inductive effect, hence it is least acidic among the following.
Why p-nitrophenol is more acidic than Ortho Nitrophenol?
Due to intramolecular hydrogen bonding, hydrogen is not easily daunted by o-nitrophenol whereas in p-nitrophenol the hydrogen free so, p-nitrophenol can donate proton easily hence p-nitrophenol is more acidic than o-nitrophenol.
Which of the following fact's explain as to why p-nitrophenol is more acidic than phenol?
The p- nitrophenol is more acidic than phenol because the deactivating nature of nitro group and after loosing the protons it makes more resonance stabilization as nitro group is having strong deactivating nature so it attracts electrons towards it.
What is the color of p-nitrophenol?
IndicatorColor at pH incidatedBelow 6Above 9p-nitrophenolColorlessYellowPhenolphthaleinColorlessPink(mixture of phenolphthalein and p-nitrophenol)ColorlessRed
Why p-nitrophenol is more soluble than O-nitrophenol in water?
Due to this H-bonding, the o-isomer of nitrophenol is not able to form H-bonds with solvent molecules. This makes it less soluble in water in comparison with p-nitrophenol. Therefore, we can conclude that o-nitrophenol forms intramolecular H-bonding which is the reason for its low solubility.
What is PnP chemical?
Poly-Solv PnP – Propylene Glycol n-Propyl Ether is a clear, colorless liquid having a mild characteristic odor.
What is the role of P nitrophenol pNP in the enzyme assay?
p-Nitrophenyl Phosphate (PNPP) is a non-proteinaceous, non-specific substrate used to assay protein, alkaline and acid phosphatases. … The assay can be used for the quick analysis of the protein phosphatase activity under any non-standard conditions.
How does pH affect alkaline phosphatase?
The pH optimum for alkaline phosphatase activity was 9.0 for all of the isolates tested. The pH optimum for acid phosphatase activity was 5.8 for 67% of the isolates and 4.8 for 33%.
What reaction is catalyzed by the enzyme alkaline phosphatase?
Alkaline phosphatase catalyzes the hydrolysis of glucose-6-phosphate via a dissociative mechanism | Journal of the American Chemical Society.
What is the difference between Bathochromic and Hypsochromic shift?
Bathochromic: a shift of a band to lower energy or longer wavelength (often called a red shift). Hypsochromic: a shift of a band to higher energy or shorter wavelength (often called a blue shift).
What shows bathochromic shift in polar solvent?
We find that the specific effect of the polar solvent, which manifests itself here as a bathochromic shift of one of π-π* bands, is caused by the formation of hydrogen bonds between solvent molecules and the molecule under study and, as a consequence, by a decrease in the energy gap between the corresponding occupied ( …
What causes red shift in UV spectroscopy?
Auxochrome: A substituent on a chromophore that leads to a red shift. Blue Shift or Hypsochromic Effect: A change in absorbance that leads to a shorter wavelength. λmax: The “top” (i.e., point of maximum absorbance) of any absorbance peak in a UV or VIS spectrum. Hypochromic Effect: A decreased absorption intensity.
What causes red shifting?
Red shift occurs due to the Doppler effect, which says that the wavelength of light changes depending on if the wave source is moving toward or away from the detector, with the detector being the person who experiences the wave.