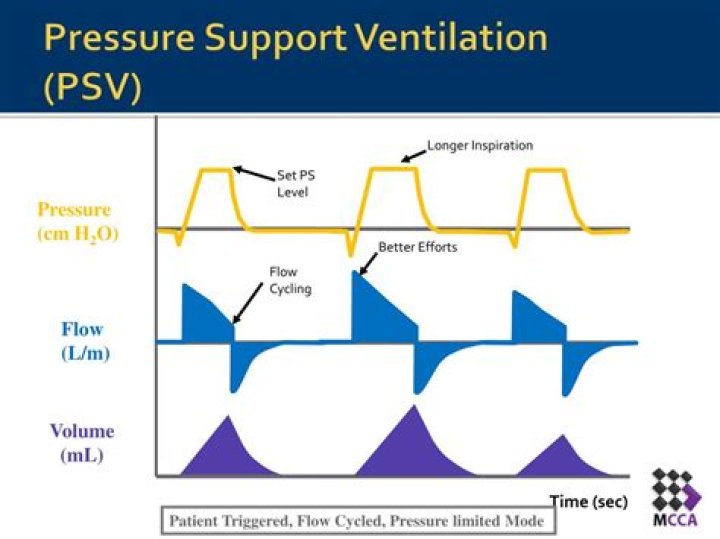
Soda lime is a mixture of NaOH & CaO chemicals, used in granular form in closed breathing environments, such as general anaesthesia, submarines, rebreathers and recompression chambers, to remove carbon dioxide from breathing gases to prevent CO2 retention and carbon dioxide poisoning.
When do you change soda lime in Anaesthesia machine?
For a standard anesthesia machine, Dispomed recommends changing the soda lime after 14 hours of use. However, keep in mind that soda lime may be exhausted faster than in 14 hours and that you may have to change it more often than every 14 hours.
Does soda lime absorb oxygen?
Closed Environments. Soda lime’s proficiency at removing carbon dioxide from air, leaving the oxygen behind, makes it a crucial part of CO2 “scrubbers,” or rebreathing systems.
What is the function of soda lime in respiration?
Soda lime is used to absorb any carbon dioxide produced by the respiring organisms. The gas volume will reduce as oxygen is removed for respiration.Does soda lime absorb isoflurane?
Normal (hydrated) soda lime absorbent (approximately 95% calcium hydroxide [Ca(OH)2], the remaining 5% consisting of a mixture of sodium hydroxide [NaOH] and potassium hydroxide [KOH]) degrades sevoflurane to the nephrotoxin Compound A, and desiccated soda lime degrades desflurane, enflurane, and isoflurane to carbon …
How much CO2 does soda lime absorb?
Soda lime absorbs about 19% of its weight in carbon dioxide, hence 100 g of soda lime can absorb approximately 26 L of carbon dioxide. The first neutralization reaction involves the formation of carbonic acid from CO2 and water.
What is the pH of soda lime?
Soda lime consists of 94% calcium hydroxide and 5% sodium hydroxide with a small amount of potassium hydroxide (less than 0.1%). It has a pH of 13.5 and a moisture content of 14–19%. Some modern types of soda lime have no potassium hydroxide.
Why is soda lime used in the preparation of methane?
Answer: Sodalime instead of caustic soda is taken in the laboratory preparation of methane gas from Sodium acetate . Caustic soda or sodium hydroxide absorbs water from the atmosphere and thus sodalime that is sodium hydroxide mixed with calcium oxide is stable and absorbs less water or moisture.Why is soda lime placed in the tube?
(b) It is the property of limewater to absorb carbon dioxide. Hence it is placed in the flask, so as to absorb carbon dioxide and turn milky in the presence of any carbon dioxide.
What is chemical composition of soda lime?Soda Lime Composition Calcium oxide, CaO (about 75%) Water, H2O (about 20%) Sodium hydroxide, NaOH (about 3%) Potassium hydroxide, KOH (about 1%).
Article first time published onIs soda lime hazardous?
Ingestion: Causes gastrointestinal tract burns. Causes severe pain, nausea, vomiting, diarrhea, and shock. Inhalation: Irritation may lead to chemical pneumonitis and pulmonary edema. Causes severe irritation of upper respiratory tract with coughing, burns, breathing difficulty, and possible coma.
Does soda lime dissolve in water?
Soda lime, solid is generally a white to grayish white colored solid. It is the mixture of calcium hydroxide and sodium or potassium hydroxide, both corrosive materials. It is noncombustible and soluble in water with release of heat.
What is the soda lime test?
Soda lime test: A small amount of an organic compound is heated strongly with soda lime (CaO + NaOH). The liberation of ammonia indicates the presence of nitrogen in the organic compound. Limitation: This test is not given by all nitrogenous compounds. Nitro and azo compounds do not respond to this test. 864 Views.
What is desiccated absorbent?
Desiccated carbon dioxide absorbents degrade desflurane, enflurane, and isoflurane to carbon monoxide (CO) in vitro and in anesthesia machines, which can result in significant clinical CO exposure. … Complete rehydration also decreased CO formation from enflurane and isoflurane to undetectable concentrations.
Why is soda lime pink?
Sofnolime 2550 pink to white USP is a medical grade soda lime used to remove carbon dioxide in closed anesthesia circuits. The pink to white colour indicator changes the granules from pink to white when carbon dioxide is absorbed.
Is soda glass safe?
Untreated soda-lime glass is far more likely than the others to break from a tumble off your table. It’s also more susceptible to breaking from thermal shock (sudden or extreme changes in temperature, such as what happens when you pour a hot liquid into a cold glass).
What is the human blood pH?
A pH of 7.0, in the middle of this scale, is neutral. Blood is normally slightly basic, with a normal pH range of about 7.35 to 7.45. Usually the body maintains the pH of blood close to 7.40.
Does CO2 react with soda lime?
The Chemistry: Carbon dioxide and the water contained in the soda lime react to form carbonic acid: CO2 + H2O = H2CO3.
What is the water content of soda lime?
Fresh soda lime contains 15–20% water, which provides the maximum CO2absorption capacity and rate of CO2absorption.
Does lime absorb CO2?
As the mortar hardens, it reabsorbs the carbon dioxide in the atmosphere and eventually re-hardens into limestone. Through this cycle, lime is widely considered a carbon-neutral building material, as it reabsorbs the carbon dioxide released during the manufacturing process.
What is the use of lime water?
Drinking lime water improves digestion. Limes are acidic and they help saliva break down food for better digestion. Additionally, flavonoids in limes stimulate secretion of digestive juices. If you experience constipation, the acidity of limes can clear the excretory system and stimulate bowel activity.
Which is the chemical used to absorb oxygen?
Pyrogallic acid is the chemical which absorbs oxygen.
What is Anoxybiotic respiration?
Anaerobic respiration (anoxybiotic) is the release of energy from the incomplete breakdown of glucose in the absence of oxygen to ethanol and carbon dioxide e.g., yeast, some bacteria, muscle cells.
What is caustic soda?
Caustic soda is the chemical compound sodium hydroxide (NaOH). This compound is an alkali – a type of base that can neutralize acids and is soluble in water. Today caustic soda can be manufactured in the form of pellets, flakes, powders, solutions and more.
How is methane gas produced in the laboratory?
Methane gas can be made in the laboratory by heating a mixture of anhydrous sodium acetate and soda lime. In this mixture sodium acetate and soda lime are taken in about 1:2 ratio. Soda lime is made by heating one part of caustic soda and three parts of quenched lime. It contains NaOH and CaO.
Is soda lime a Deliquescent?
The size of the soda lime granules is 4-8 mesh (i.e. In chemical reactions, soda-lime acts as sodium hydroxide but, unlike sodium hydroxide, it is not deliquescent and does not attack glass.
What happens when sodium is heated with soda lime?
Sodium acetate in the presence of soda lime and heat, leads to the formation of methane gas and sodium carbonate. This type of reaction is called as decarboxylation reaction and as the name suggests there is a loss of a molecule of carbon dioxide from sodium acetate.
How do you dispose of soda lime?
Disposal: Reuse soda lime multiple times until it turns a violet color. Follow your school’s guidelines for chemical waste disposal. If your school does not have guidelines already for disposal of chemicals, place the spent soda lime in a sealed plastic bag and place it in the garbage.
Is soda lime hygroscopic?
Soda Lime, Extra Pure, SLR, 2.5-5mm, Not Hygroscopic, Granules, Fisher Chemical™
What is soda lime reaction?
The reaction mechanism of carbon dioxide with soda lime can be decomposed in three elementary steps: 1) CO2(g) → CO2(aq) (CO2 dissolves in water – slow and rate-determining) 2) CO2(aq) + NaOH → NaHCO3 (bicarbonate formation at high pH) 3) NaHCO3 + CaO→ CaCO3 + NaOH (NaOH recycled to step 2 – hence a catalyst)
Can soda lime be reused?
It is possible to recycle and reuse soda lime in different forms; however, the calcination process would require relatively high temperatures. On the other hand, high temperatures would ensure the biological neutrality of recycled soda lime.