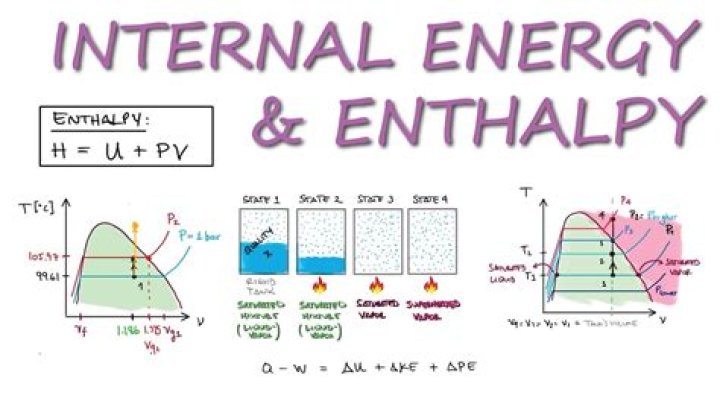
In symbols, the enthalpy, H, equals the sum of the internal energy, E, and the product of the pressure, P, and volume, V, of the system: H = E + PV. According to the law of energy conservation, the change in internal energy is equal to the heat transferred to, less the work done by, the system.
Is internal energy and enthalpy the same thing?
The main difference between enthalpy and internal energy is that enthalpy is the heat absorbed or evolved during chemical reactions that occur in a system whereas internal energy is the sum of potential and kinetic energy in a system.
What do you mean by internal energy and enthalpy?
0. The internal energy of a system is simply the sum of its potential and kinetic energies i.e. E(int) = U + K. The enthalpy is then the total internal energy of the system as well as how much pressure the system exerts on the volume it inhabits i.e.
Is internal energy and enthalpy path dependent?
The change in internal energy and change in heat enthalpy does not depend upon the path by which changes are brought in.What is difference between energy and enthalpy?
What is the difference between Energy and Enthalpy? Energy is only measured in joules, but enthalpy is measured in both joules and joules per mole. Enthalpy is also a form of energy. Energy is a state of the matter, but enthalpy is always the energy change between two states.
Why both internal energy and enthalpy are state functions?
Both enthalpy and the internal energy are often described as state functions. … It means that when a system undergoes any change whatever, then the alteration in its enthalpy (or its internal energy) depends only on the initial state of the system and its final state.
Why is internal energy less than enthalpy?
Why is enthalpy greater than internal energy? Internal energy is a heat of reaction at a constant volume where enthalpy is a heat of reaction plus pressure-volume work at constant pressure. This additional factor of pressure-volume work makes the enthalpy of an ideal gas 40% greater than its internal energy.
Is enthalpy path dependent?
Enthalpy is a state function because it depends only on two thermodynamic properties of the state the substance is at the moment (like temperature and pressure, or temperature and entropy, or any pair of other state functions). It does not depend on the path followed by the substance to get there.Does internal energy depend on the path?
The internal energy of a system of constant composition can be changed by work or heat interactions with its surroundings. Both of these energy transfer processes are path dependent, however, the internal energy is a function only of the state of the system.
How is enthalpy related to energy?In symbols, the enthalpy, H, equals the sum of the internal energy, E, and the product of the pressure, P, and volume, V, of the system: H = E + PV. According to the law of energy conservation, the change in internal energy is equal to the heat transferred to, less the work done by, the system.
Article first time published onWhat determines internal energy?
The internal energy of a system depends on its entropy S, its volume V and its number of massive particles: U(S,V,{Nj}). It expresses the thermodynamics of a system in the energy representation.
What affects internal energy?
The internal energy can be altered by modifying the object’s temperature or volume without altering the amount of particles inside the body. Temperature: As a system’s temperature increases, the molecules will move faster, thus have more kinetic energy and thus the internal energy will increase.
Why is enthalpy considered more useful than internal energy?
WHY ENTHALPY IS CONSIDERED MORE USEFUL THAN INTERNAL ENERGY IN CHEMICAL REACTIONS? … ΔH is equal to the change in the internal energy of the system, plus the work that the system has done on its surroundings. So, enthalpy term is used in place of internal energy because it gives more precise value of energy.
What is difference between enthalpy and free energy?
The key difference between free energy and enthalpy is that free energy gives the total energy available to perform thermodynamic work whereas enthalpy gives the total energy of a system that can be converted to heat.
What is the difference between change in enthalpy and change in internal energy at constant volume?
Thus, the difference between enthalpy and internal energy arises under constant pressure – the difference is the pressure-volume work. … In fact, in chemistry, reactions under constant pressure are much more common than reactions under constant volume.
What is the difference between the internal energy and enthalpy for one mole of ideal gas?
Previously, we said that the enthalpy of an ideal gas is independent of pressure at constant temperature. And the internal energy of an ideal gas is independent of volume at constant temperature.
Why do we generally study enthalpy changes and not internal energy changes?
Thermodynamics. Why heat changes reported are usually enthalpy changes and not internal energy? This is because most of the processes are carried out in open vessels i.e. at constant pressure. … Calculate enthalpy change for the reaction at 298 K.
Does enthalpy increase in an endothermic reaction?
Endothermic reaction: In an endothermic reaction, the products are higher in energy than the reactants. Therefore, the change in enthalpy is positive, and heat is absorbed from the surroundings by the reaction.
What is the formula for change in internal energy?
The first law of thermodynamics states that the change in internal energy of a system equals the net heat transfer into the system minus the net work done by the system. In equation form, the first law of thermodynamics is ΔU = Q − W. Here ΔU is the change in internal energy U of the system.
Is enthalpy extensive or intensive?
Intensive properties are properties that do not depend on the quantity of matter. For example, pressure and temperature are intensive properties. Energy, volume and enthalpy are all extensive properties.
Is enthalpy change a state function?
Enthalpy is a state function. This implies that when a system changes from one state to another, the change in enthalpy is independent of the path between two states of a system.
Is internal energy equal to potential energy?
The internal energy is the total amount of kinetic energy and potential energy of all the particles in the system.
Is enthalpy a path function?
Enthalpy is considered a state function because its current value will only depend upon the final and initial values of heat in a reaction, but not the path or process that occurred for it to reach that value.
Why does internal energy only depends on temperature?
The meaning of “depend only on temperature” is that the internal energy of the gas does not change when it is allowed to expand into a vacuum. In that sense, the internal energy is independent of pressure and volume, and therefore dependent only on temperature.
Is internal energy is a state function?
Internal energy only depends upon the initial and final state of the system, not on the path followed. So, the internal energy is a state function.
Is change in enthalpy path independent?
As enthalpy is a state function, the change in enthalpy is independent of the path of change, so that , for any reaction can be calculated from a suitable combination of values for standard reference reactions.
Which function is enthalpy?
Enthalpy is a function of both temperature and pressure. It is a function of Temperature. It is given by mxCpx(delta T). So enthalpy is directly proportional to temperature.
What is meant by internal energy?
internal energy, in thermodynamics, the property or state function that defines the energy of a substance in the absence of effects due to capillarity and external electric, magnetic, and other fields.
What happens to internal energy when temperature increases?
The internal energy is the total amount of kinetic energy and potential energy of all the particles in the system. When energy is given to raise the temperature , particles speed up and they gain kinetic energy.
What is internal energy equal to?
The internal energy is equal to the heat of the system. The surrounding heat increases, so the heat of the system decreases because heat is not created nor destroyed. Therefore, heat is taken away from the system making it exothermic and negative.
What is difference between total energy and internal energy?
The internal energy of a system is identified with the random, disordered motion of molecules; the total (internal) energy in a system includes potential and kinetic energy. … It is the sum of all the microscopic energies such as: translational kinetic energy.