According to the Maxwell Boltzmann energy distribution, the most probable energy is Ep=kT2.
What is the most probable kinetic energy?
Most probable kinetic energy is the energy possessed by the maximum number of reactant molecules at a temperature T, and it is shown as a peak on the Maxwell-Boltzmann energy distribution graph (fraction of molecules versus Kinetic energy).
What is Boltzmann probability distribution?
In statistical mechanics and mathematics, a Boltzmann distribution (also called Gibbs distribution) is a probability distribution or probability measure that gives the probability that a system will be in a certain state as a function of that state’s energy and the temperature of the system.
What does the Maxwell Boltzmann distribution show energy?
The Maxwell–Boltzmann distribution describes the distribution of speeds among the particles in a sample of gas at a given temperature. The distribution is often represented graphically, with particle speed on the x-axis and relative number of particles on the y-axis. Created by Sal Khan.What is the condition for most probable distribution?
The most probable distribution in Boltzman theory is extremely sharp for large number of particles. ⟨O⟩=∫ρ({ϵi:ai})O({ϵi:ai})d{ϵi:ai}. If ρ({ϵi:ai}) is a delta function distribution, only the most probable distribution, ρmost probable is need to calculate observables.
What is Boltzmann distribution law?
∎ The Boltzmann distribution law states that the. probability of finding the molecule in a particular. energy state varies exponentially as the energy. divided by k. B.
What is the most probable kinetic energy corresponding to Maxwellian velocity distribution?
According to the Maxwell Boltzmann energy distribution, the most probable energy is Ep=kT2.
What does the Boltzmann equation measure?
The Boltzmann equation can be used to determine how physical quantities change, such as heat energy and momentum, when a fluid is in transport. … See also convection–diffusion equation.What does a Boltzmann distribution show?
The Boltzmann distribution (AKA kinetic energy distribution) is a distribution that describes the amount of kinetic energy that a proportion of gas particles have in a given sample.
What is the Maxwell-Boltzmann theory?The Maxwell-Boltzmann equation, which forms the basis of the kinetic theory of gases, defines the distribution of speeds for a gas at a certain temperature. From this distribution function, the most probable speed, the average speed, and the root-mean-square speed can be derived.
Article first time published onIs Boltzmann distribution a normal distribution?
In the normal distribution, the probability that an atom will have a given energy decreases exponentially as the energy rises. The bell-like Maxwell-Boltzmann distribution is derived from the exponential decay of the number of particles with a given energy. … The result is a bell-like distribution.
What is Boltzmann gas?
At the microscopic level, the temperature—according to Ludwig Boltzmann—is a measure of the kinetic energy associated with the disordered motion of the atoms or molecules that constitute the sample. …
What is meant by most probable distribution?
“Most probable” refers to having a large number of different ways of achieving the distribution. For example, in a solution, the solute molecules are typically equally distributed throughout the solution volume.
What is the most probable macrostate?
is the number of microstates accessible to a system when it has an energy and is the number of microstates accessible to the system when it has an energy .
What do microstates do with Boltzmann distribution?
The probability that case 10 is the case is 420 in 1716 (the highest of all). For a large number N (for example, 6.02 x 1023) and a correspondingly increased total energy E: The number of microstates in the most probable distribution far outweighs the other distributions.
What is the most probable velocity?
Most probable velocity is the velocity possessed by maximum fraction of molecules at the same temperature.
What is the most probable velocity for a one dimensional gas velocity distribution?
The most probable velocity for a 1-dimensional gas velocity is the same as 3 dimensions. This is known because of the differentiation of the Maxwell-Boltzmann distribution and setting the result equal to zero.
What happens to the most probable kinetic energy?
Answer: Kinetic energy is directly proportional to the absolute temperature and the number of molecules possessing higher energies increases with increase in temperature, i.e., most probable kinetic energy increases with increase in temperature.
What is Boltzmann distribution of energy?
The Maxwell–Boltzmann distribution concerns the distribution of an amount of energy between identical but distinguishable particles. It represents the probability for the distribution of the states in a system having different energies. A special case is the so-called Maxwell distribution law of molecular velocities.
What does Boltzmann factor represent?
Boltzmann’s factor is e-E/kT, which expresses the “probability” of a state of energy E relative to the probability of a state of zero energy. This factor can be used to introduce temperature into a wide variety of physical problems, and is often taken as a starting point.
What type of distribution is the Maxwell Boltzmann distribution?
A Maxwell-Boltzmann Distribution is a probability distribution used for describing the speeds of various particles within a stationary container at a specific temperature. The distribution is often represented with a graph, with the y-axis defined as the number of molecules and the x-axis defined as the speed.
What is the Boltzmann constant in eV?
Click symbol for equationBoltzmann constant in eV/KNumerical value8.617 333 262… x 10-5 eV K-1Standard uncertainty(exact)Relative standard uncertainty(exact)
What is Boltzmann constant in chemistry?
The Boltzmann constant (k or kB) is the physical constant relating temperature to energy. It is named after the Austrian physicist Ludwig Eduard Boltzmann. Its experimentally determined value (in SI units, 2002 CODATA value) is: kB=1.3806505(24)×10−23JK−1.
What is Boltzmann transport equation in solid state physics?
When electric or magnetic fields are present charge will be pushed around inside a solid. We define a probability density function f(→r,→k,t) that decribes the probability of finding an electron at position →r with a wave vector →k at time t.
What are the basic approaches of Maxwell Boltzmann statistics?
In statistical mechanics, Maxwell–Boltzmann statistics describes the distribution of classical material particles over various energy states in thermal equilibrium. It is applicable when the temperature is high enough or the particle density is low enough to render quantum effects negligible.
Why is the Boltzmann approximation used?
Fermi Dirac Distribution Boltzmann Approximation Maxwell- Boltzmann distribution is the commonly used Fermi Dirac distribution approximation. When the difference between the carrier’s energy and Fermi level is large compared to, the term 1 in the denominator can be neglected.
What is most probable speed of gas molecules?
The speed that corresponds to the peak of the curve is called the most probable speed. More molecules travel at (or close to) this speed than any other. For N2 at 25ºC, the most probable speed is 421 m/sec.
What is Maxwell Boltzmann distribution used for?
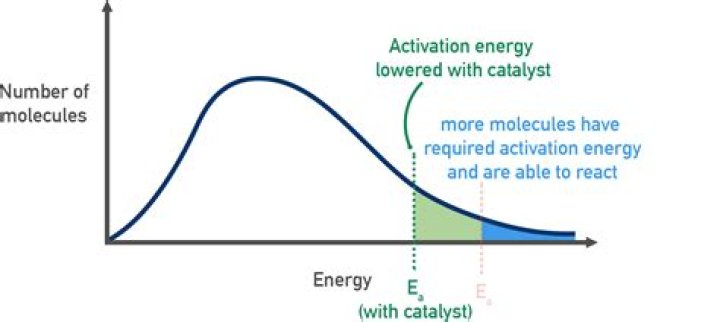
The Maxwell–Boltzmann distribution (Figure 3) can be used to describe the fraction of molecules with different kinetic energies in a population of molecules at a specified temperature, and allows the estimation of the fraction of the molecules in a population that exceed the activation energy of a reaction.
What is Boltzmann population ratio?
The Boltzmann distribution law is then given by the expression. This expression states that the natural logrithm of the ratio of the number of particles in the upper state to the number in the lower state is equal to the negative of their energy separation divided by kT.
What are classical statistics?
Classical statistics uses techniques such as Ordinary Least Squares and Maximum Likelihood – this is the conventional type of statistics that you see in most textbooks covering estimation, regression, hypothesis testing, confidence intervals, etc. … In fact Bayesian statistics is all about probability calculations!
Does deposition increase entropy?
Consequently, for any substance, Sgas > Sliquid > Ssolid, and the processes of vaporization and sublimation likewise involve increases in entropy, ΔS > 0. Likewise, the reciprocal phase transitions, condensation and deposition, involve decreases in entropy, ΔS < 0.