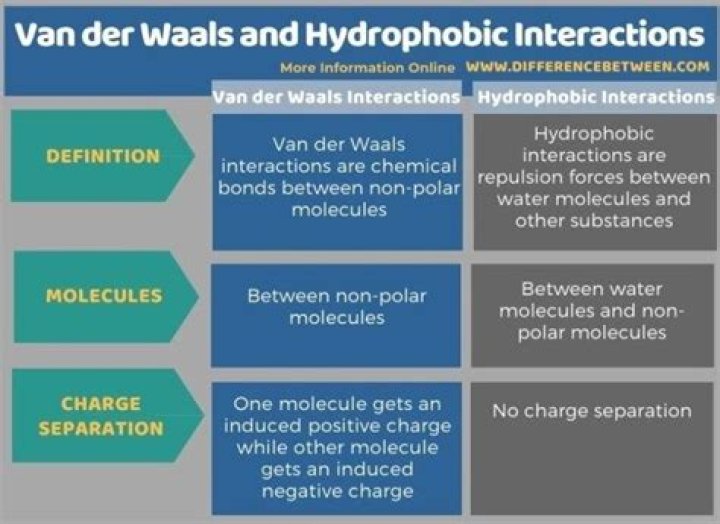
The key difference between Van der Waals and hydrophobic interactions is that Van der Waals interactions are attraction forces between non-polar molecules, whereas hydrophobic interactions are repulsion forces between water molecules and other molecules.
Is hydrophobic stronger than Van der Waals?
Hydrophobic interactions occur when nonpolar (hydrophobic) amino acids associate with each other and cluster together to hide from water, usually on the inside of a protein. … Hydrophobic interactions are also weak, but stronger than Van der Waal’s forces.
What is the difference between Van der Waals and hydrogen bonding?
Hydrogen bonds occur in inorganic molecules, such as water, and organic molecules, such as DNA and proteins. Van der Waals attractions can occur between any two or more molecules and are dependent on slight fluctuations of the electron densities.
What type of interaction is van der Waals?
A van der Waals interaction is a relatively weak force ranging from 0.5 to 1 kcal/mol and is nonionic in nature. Neutral molecules containing electronegative atoms, like oxygen and nitrogen, have a tendency to draw the electron cloud toward itself through the covalent bond from its less electronegative neighbor atom.Is hydrophobic interaction intermolecular forces?
Hydrophobic interactions is one major type of intermolecular force that plays a vital role in many life processes in Chemistry and Physics.
Do hydrophobic interactions increase entropy?
Entropy increases because water molecules exclude the nonpolar solute in order to interact with each other and regain a higher state of disorder. Explanation: This is called the hydrophobic effect.
What's the relationship between Van der Waals forces and hydrophobic interactions?
The relationship between van der waals forces and hydrophobic interactions is that the van der waals act to bind the hydrophobe – non-polar substance – together, to separate from the polar solvent/water, and these contribute to the energy needed to separate the two substances.
What is the difference between van der Waals and London dispersion?
Glossary. London dispersion forces: The intermolecular forces that occur between atoms and between nonpolar molecules as a result of the motion of electrons. Van der Waals forces: The weakest intermolecular force and consist of dipole-dipole forces and dispersion forces .What is the difference between dipole dipole and van der Waals?
Dipole-dipole force are a type of Van Der Waals force. When two polar molecules interact, opposite partial charges attract, similarly to ionic bonding, but generally weaker, because of the smaller charge magnitude. … Two simple polar molecules with a dipole-dipole interaction shown as a dotted line.
What are dipole interactions?Dipole-Dipole interactions result when two dipolar molecules interact with each other through space. When this occurs, the partially negative portion of one of the polar molecules is attracted to the partially positive portion of the second polar molecule.
Article first time published onAre hydrogen bonds van der Waals interactions?
Hydrogen bonding is the third type of van der Waals’ forces. It is exactly the same as dipole-dipole interaction, it just gets a special name. A hydrogen bond is a dipole dipole interaction that occurs between any molecule with a bond between a hydrogen atom and any of oxygen/fluorine/nitrogen.
Which is stronger hydrogen bonds or van der Waals interactions?
The hydrogen bond that was generally from 5 to 30 kJ /mol is stronger than a van der Waals interaction, but weaker than covalent or ionic bonds.
Why are hydrogen bonds and van der Waals interactions necessary for?
Why are hydrogen bonds and van der Waals interactions necessary for cells? Hydrogen bonds and van der Waals interactions form weak associations between different molecules. They provide the structure and shape necessary for proteins and DNA within cells so that they function properly.
How do hydrophobic and hydrophilic molecules interact with each other?
Nonpolar molecules that repel the water molecules are said to be hydrophobic; molecules forming ionic or a hydrogen bond with the water molecule are said to be hydrophilic. … One end of the organic molecule binds to the solid surface via a covalent bond while the other end points outwards.
Are hydrophobic interactions stronger than ionic interactions?
Ionic bonds are less strong – although some are very close to covalent, whereas others are quite weak. Hydrophobic (lipophilic) and hydrophilic bonds are much weaker and hydrogen bonds are weakest. BUT… Covalent bonds are strongest.
What is hydrophobic interaction in protein?
A Hydrophobic Effect. The major driving force in protein folding is the hydrophobic effect. This is the tendency for hydrophobic molecules to isolate themselves from contact with water. As a consequence during protein folding the hydrophobic side chains become buried in the interior of the protein.
Is hydrophobic effect van der Waals?
Summary. It has been shown thermodynamically, and illustrated by means of a typical example, that the preferential attraction between hydrophobic determinants immersed in water, commonly alluded to as the “hydrophobic effect”, can be entirely ascribed to van der Waals interactions.
What do hydrophobic amino acids interact with?
Hydrophobic bonds in proteins arise as a consequence of the interaction of their hydrophobic (i.e., “water-disliking”) amino acids with the polar solvent, water. The hydrophobic amino acids are gly, ala, val, leu, ile, met, pro, phe, trp (see amino acid structures for reference).
How does water affect van der Waals forces?
Water molecules in liquid water are attracted to each other by electrostatic forces, and these forces have been described as van der Waals forces or van der Waals bonds. … Even though the molecules don’t have permanent dipole moments, they can have instantaneous dipole moments which change or oscillate with time.
What is the difference between hydrophilic and hydrophobic substances?
Materials with a special affinity for water — those it spreads across, maximizing contact — are known as hydrophilic. Those that naturally repel water, causing droplets to form, are known as hydrophobic.
Why are hydrophobic interactions entropy driven?
The hydrophobic effect was found to be entropy-driven at room temperature because of the reduced mobility of water molecules in the solvation shell of the non-polar solute; however, the enthalpic component of transfer energy was found to be favorable, meaning it strengthened water-water hydrogen bonds in the solvation …
What is the effect of hydrophobic interactions on the tertiary structure of proteins?
Tertiary Structure. Hydrophobic Interactions: Soluble globular proteins fold so that hydrophobic side chains are mostly sequestered in the core of the protein. The removal of the non-polar groups from water (i.e. the hydrophobic effect) is the primary force stabilizing tertiary structure.
What is the difference between dipole-dipole and London dispersion?
The main difference between dipole-dipole and London dispersion forces is that dipole-dipole forces occur among molecules with dipole moment whereas London dispersions occur due to instantaneous dipoles that form in atoms or nonpolar molecules.
What is the difference between intramolecular forces and intermolecular forces?
Intramolecular forces are the forces that hold atoms together within a molecule. Intermolecular forces are forces that exist between molecules.
What is difference between dipole-dipole forces and hydrogen bonding?
An ion-dipole force is a force between an ion and a polar molecule. A hydrogen bond is a dipole-dipole force and is an attraction between a slightly positive hydrogen on one molecule and a slightly negative atom on another molecule.
Why is it called London dispersion?
London dispersion force is a weak intermolecular force between two atoms or molecules in close proximity to each other. … The force gets its name because Fritz London first explained how noble gas atoms could be attracted to each other in 1930.
Are dispersion forces and London dispersion forces the same?
London dispersion forces (LDF, also known as dispersion forces, London forces, instantaneous dipole–induced dipole forces, Fluctuating Induced Dipole Bonds or loosely as van der Waals forces) are a type of force acting between atoms and molecules that are normally electrically symmetric; that is, the electrons are …
What is the meaning of van der Waals?
Definition of van der Waals forces : the relatively weak attractive forces that act on neutral atoms and molecules and that arise because of the electric polarization induced in each of the particles by the presence of other particles.
What do van der Waals forces do?
van der Waals forces, relatively weak electric forces that attract neutral molecules to one another in gases, in liquefied and solidified gases, and in almost all organic liquids and solids.
What types of molecules exhibit dipole-dipole interactions?
Dipole–dipole forces occur between molecules with permanent dipoles (i.e., polar molecules). For molecules of similar size and mass, the strength of these forces increases with increasing polarity. Polar molecules can also induce dipoles in nonpolar molecules, resulting in dipole–induced dipole forces.
Are van der Waals forces the same as intermolecular forces?
Van der Waals forces are a type of intermolecular force. They refer to London forces (electrostatic forces of attraction between temporary induced dipoles). The name is is also sometimes used to refer to forces between permanent dipoles and temporary dipoles and, rarely, to forces between two permanent dipoles (PDD).