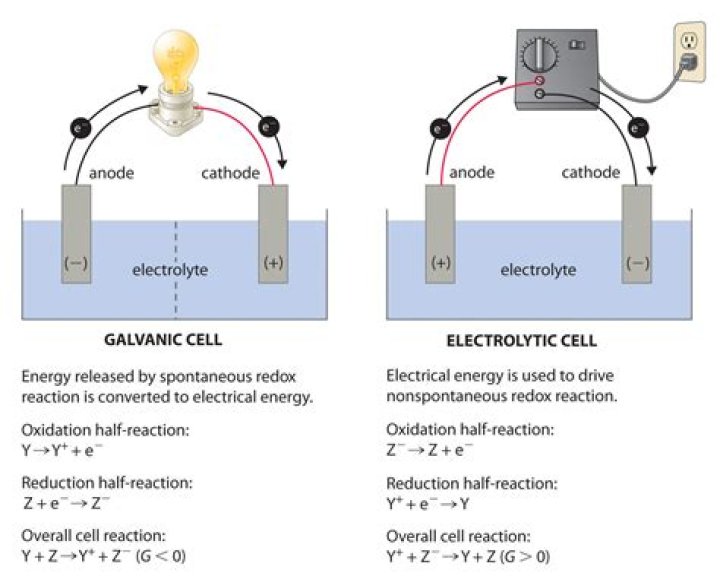
Electrons flow in the external circuit. In electrochemistry, the standard electrode potential, abbreviated E°, is the measure of the individual potential of a reversible electrode at standard state, which is with solutes at an effective concentration of 1 M, and gases at a pressure of 1 atm.
What does E mean in electrochemistry?
In electrochemistry, standard electrode potential (E°) is defined as “The value of the standard emf (electromotive force) of a cell in which molecular hydrogen under standard pressure is oxidized to solvated protons at the left-hand electrode”.
What is E * in chemistry?
E* refers to cell potential at standard conditions, it does NOT change with amount of substance. E is just the potential of the cell under ANY conditions.
What is E in the Nernst equation?
F = Faraday’s constant (96,485 C/mole e-) n = number of moles of electrons transferred in the balanced equation for the reaction occurring. in the cell. Q = reaction quotient for the reaction.What does E cell mean chemistry?
Electrons are able to move between electrodes because the chemical reaction is a redox reaction. … The cell potential (Ecell) is measured in voltage (V), which allows us to give a certain value to the cell potential.
What is the formula of E cell?
The overall cell potential can be calculated by using the equation E0cell=E0red−E0oxid. Step 2: Solve. Before adding the two reactions together, the number of electrons lost in the oxidation must equal the number of electrons gained in the reduction.
What is E Not cell in electrochemistry?
n is the number of electrons transferred in the redox reaction. … Q is the reaction quotient, i.e. not-yet-equilibrium constant. E is the “electromotive force” for the cell process.
What is E in organic chemistry?
E: Describes the configuration of a double bond in which the two groups of highest Cahn-Ingold-Prelog priority lie on opposite sides.How do you find the E cell in the Nernst equation?
- E∘cell=RTnFlnK.
- E∘cell=0.0257VnlnK=0.0592VnlogK(at298.15K)
- Ecell=E∘cell−RTnFlnQ(Nernst equation)
- Ecell=E∘cell−0.0257VnlnQ=E∘cell−0.0592VnlogQ(at298.15K)
- ΔG = −nFEcell
- ΔG∘=−nFE∘cell.
- wele=wmax=−nFEcell.
Wavelength is related to energy and frequency by E = hν = hc/λ, where E = energy, h = Planck’s constant, ν = frequency, c = the speed of light, and λ = wavelength.
Article first time published onWhat is E and E not?
E cell means electrode potential of a cell. E0 cell means standard electrode potential means electrode potential measured at 1 atmosphere pressure, 1 molar solution at 25° C.
What is the value of E cell?
The value of ${E^ \circ }_{cell}$ found is $1.52{\text{ V}}$. The answer of the given question is \[1.5055{\text{ V}}\]. Note: Nernst equation is the equation that relates half or full cell reaction to temperature concentration of the species and the standard electrode potential.
What is F in Gibbs free energy?
F = NAqe ≈ 96485 C/mol, Faraday constant (charge per mole of electrons), E, cell potential, E°, standard cell potential.
Is E cell intensive or extensive?
i ECell and ΔrG of cell reaction both are extensive properties. ii ECell and ΔrG of cell reaction both are intensive properties.
What is F in Nernst equation?
R is the ideal gas constant (joules per kelvin per mole), T is the temperature in kelvins, F is the Faraday’s constant (coulombs per mole).
What is E in redox reaction?
For standard electrochemical cells 1: A redox reaction is spontaneous if the standard electrode potential for the redox reaction, Eo(redox reaction), is positive. … If Eo(redox reaction) is negative (Eo(redox reaction) < 0), the reaction will not proceed in the forward direction (non-spontaneous).
What is 2e in chemistry?
Chemistry 2e is designed to meet the scope and sequence requirements of the two-semester general chemistry course. The textbook provides an important opportunity for students to learn the core concepts of chemistry and understand how those concepts apply to their lives and the world around them.
What is the difference between E and EO?
Eo is at standard state (25 degrees C and 1 atm) while E is the cell potential at anything other than standard state.
How do you find e?
We’ve learned that the number e is sometimes called Euler’s number and is approximately 2.71828. Like the number pi, it is an irrational number and goes on forever. The two ways to calculate this number is by calculating (1 + 1 / n)^n when n is infinity and by adding on to the series 1 + 1/1! + 1/2!
Can e cells negative?
If E°cell is negative, then the reaction is not spontaneous under standard conditions, although it will proceed spontaneously in the opposite direction. The answer therefore is: The reaction as written is nonspontaneous under standard conditions.
What is the difference between Ecell and E cell?
Eocell is the standard state cell potential, meaning the condition of [1M] for solutes or 1 bar for gases is met. Ecell is the cell potential at non standard state conditions. You can use the Nernst equation to find Ecell using Eocell.
What is the EMF of standard hydrogen electrode?
Its absolute electrode potential is estimated to be 4.44 ± 0.02 V at 25 °C, but to form a basis for comparison with all other electroreactions, hydrogen’s standard electrode potential (E°) is declared to be zero volts at any temperature.
Does E cell depend on concentration?
An electrochemical cell of this type, in which the anode and cathode compartments are identical except for the concentration of a reactant, is called a concentration cell. As the reaction proceeds, the difference between the concentrations of Ag+ in the two compartments will decrease, as will Ecell.
What is E and Z method of nomenclature?
E–Z configuration, or the E–Z convention, is the IUPAC preferred method of describing the absolute stereochemistry of double bonds in organic chemistry.
How do you know if Z or E?
If the compound contains more than one double bond, then each one is analyzed and declared to be E or Z. The configuration at the left hand double bond is E; at the right hand double bond it is Z.
What is the difference between E and Z isomers?
The difference between E and Z isomers is that the E isomers have the substituents with higher priority in the opposite sides whereas the Z isomers have the substituents with higher priority on the same side.
What is energy measured in E hv?
Energy in E=hv is the energy in a wave of frequency v. This also represents the fact that energy is quantized and comes in small packets, each packet having an energy given by hv. High frequency photons have higher energy and vice versa.
Who wrote E HF?
The equation, E=hf, is referred to as the Planck relation or the Planck-Einstein relation. The letter h is named after Planck, as Planck’s constant.
What is E negative in chemistry?
This negative sign means that the energy of the electron in the atom is lower than the energy of a free electron at rest. A free electron at rest means that is infinitely far away from the nucleus and has the negligible attraction towards the nucleus.
What is the value of E at 298 K?
E c e l l = 1.50553 V.
What is the value of E cell CR cr3?
The value of E°cell for Cr|Cr 3+||Hg 2 2+|Hg(l) is 1.78 V. Calculate ΔG° for this reaction at 25 . Q.