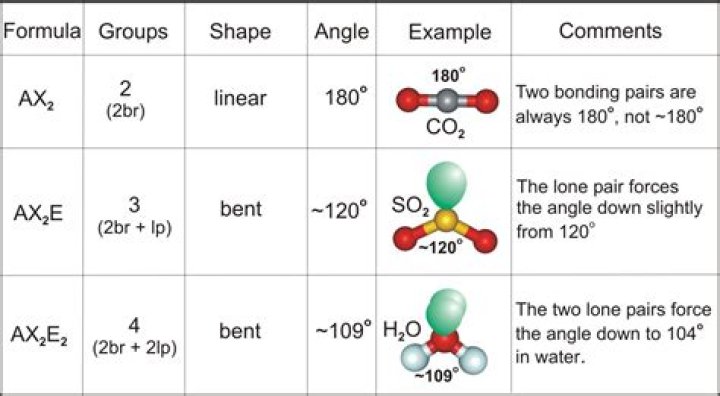
In chemistry, molecules with a non-collinear arrangement of two adjacent bonds have bent molecular geometry, also known as angular or V-shaped. Certain atoms, such as oxygen, will almost always set their two (or more) covalent bonds in non-collinear directions due to their electron configuration.
What electron geometry is bent?
# of bonding pair/s of electron on ‘central’ atom# of lone pair of electrons on ‘central’ atomMolecular Geometry30trigonal planar21bent40tetrahedral31trigonal pyramidal
What causes the bent shape in chemistry?
The reason water has a bent shape is that the two lone pair of electrons are on the same side of the molecule. … This repulsion of the lone pairs of electrons on the oxygen atom causes the bond of the hydrogen to the oxygen to be pushed downward (or upward, depending on your point of view).
How do you know if molecular geometry is bent?
If all electrons pairs are acting as bonding electrons, leaving no lone pair on the central atom (AX2), then the molecule will be linear (eg; CO2). If, however, there is one or two lone pairs (E) on the central atom, then the molecule will be bent (AX2E eg; SO2, AX2E2 eg; H2O).What is a bent bond angle?
In bent molecules, the bond angle is slightly less than 120∘ . This is because lone pairs take up more room than single bonds do. Therefore, the lone pair in a bent molecule takes up more room than the 3rd bond in a trigonal planar molecule does, thereby reducing the angle to slightly less than 120∘ .
Is bent tetrahedral?
NOTES: This molecule is made up of 4 equally spaced sp3 hybrid orbitals forming bond angles of approximately 109.5o. The shape of the orbitals is tetrahedral. Two of the orbitals contain lone pairs of electrons. The two atoms connected to the central atom form a molecule with a bent shape.
Does co2 have a bent shape?
In the carbon dioxide, the two double bonds try to get as far apart as possible, and so the molecule is linear. … To minimise repulsions, the double bonds and the lone pair get as far apart as possible, and so the molecule is bent.
Is h20 linear or bent?
H2O has four regions of electron density and two lone pairs on the central O atom so the molecule is bent.What is the difference between linear and bent?
The key difference between linear and bent molecules is that linear molecules have atoms bonded to each other, forming a straight molecule, whereas bent molecules have the atoms arranged in a bend-shape with an angle.
Is a bent shape polar or nonpolar?Formula3D Structure Shape Polarity1.CH4tetrahedral nonpolar2.NH3trigonal pyramidal polar3.H2Obent polar4.H3O+trigonal pyramidal charged
Article first time published onDoes H2S have a bent geometry?
H2S molecular geometry is bent. H2S electron geometry is tetrahedral. The total valence electron available for drawing the lewis structure of H2S is 8. The lone pair in on the central atom is 2 according to the lewis structure of SH2/H2S.
Is a bent molecule planar?
Bent or V-shaped , (trigonal/square/Pentagonal) planar, T-shaped type molecules are always Planar. A+B2 where A and B are two different elements so this type of molecule will always be Planar whatever it’s hybridization is that doesn’t matter.
Is o3 molecule bent?
Ozone has three electron groups around the central oxygen, so has the trigonal planar electron geometry. The molecular geometry is bent: The Effect of Lone Pairs (contd.)
Does SO2 have bent geometry?
While both water and sulfur dioxide molecules have a bent geometry. The bond angle in water is smaller (109.5) and SO2(120). ( 109.5 ) and S O 2 ( 120 ) .
Does H2O have bent structure?
Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. Because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). The molecule adopts a bent structure because of the two lone pairs of electrons on the oxygen atom. …
What has bent shape?
Bent molecular geometryCoordination number2Bond angle(s)90°<θ<120°μ (Polarity)>0
Why is this molecule bent instead of linear?
Linear = is just a line of atoms with a 180° angle. Notice that it’s 2 or 3 atoms total. Bent = Linear but bent due to the Lone Pairs that it contains, the more Lone Pairs the greater the bent and the smaller the degree. It is 3 atoms total.
Why is CO2 linear and H2O bent?
CO2 has two regions of electron density and no lone pairs on the central C atom so by VSEPR Theoey, the molecule is linear. H2O has four regions of electron density and two lone pairs on the central O atom so the molecule is bent. Hope this helps!
Is tetrahedral bent polar?
A tetrahedral shape is a nonpolar shape: the terminal atoms are distributed in a way that the molecule has no imbalance in polarity based on the shape alone.
What is the difference between tetrahedral bent and trigonal planar bent?
Trigonal planar: triangular and in one plane, with bond angles of 120°. Tetrahedral: four bonds on one central atom with bond angles of 109.5°.
Is NO2 bent or linear?
NO2 is a bent molecule; however, when you remove an electron from it, making it NO2+, the molecule becomes linear due to the loss of a lone electron. In NO2+, there is no repulsion taking place between the two O atoms and the lone electron on the central atom.
Why is h2s a bent?
The two regions with the bonds to hydrogen H-S-H and then the two sets of unpaired electrons. Because it has 2 bonds and 2 unshared pairs in the central atoms it would be bent.
Why is ozone bent?
AB2E: Ozone (O3) Molecules with three electron pairs have a domain geometry that is trigonal planar. Here, the lone pair on the central atom repels the electrons in the two bonds, causing the atom to adopt a bent molecular geometry.
What is the shape of CO2?
The initial VSEPR shape for the CO2 molecule is Tetrahedral. For each multiple bond (double/triple bond), subtract one electron from the final total. The CO2 molecule has 2 double bonds so minus 2 electrons from the final total.
Is n20 linear?
Nitrous oxide is linear.
Is H2S linear or bent?
H2S is a non-linear molecule. The bond angle between the two H-S bonds is about 92%. Each two 3p-orbitals of sulphur containing one electron can overlap with 1s orbitals of hydrogen atoms.
Is SiO2 bent?
So, due to repulsion between the lone pair and bond pairs the bonds are shifted slightly away from the lone pair, thus in retrospect they are shifted towards each other. Hence the linear geometry is disturbed to give a bent shape to SiO2 molecule.
Is SO3 bent?
SO3 Molecular Geometry And Bond Angles If we look at the SO3 molecular geometry it is trigonal planar with symmetric charge distribution around the central atom. Sulphur trioxide is also non-polar. It has a bond angle of 120o.