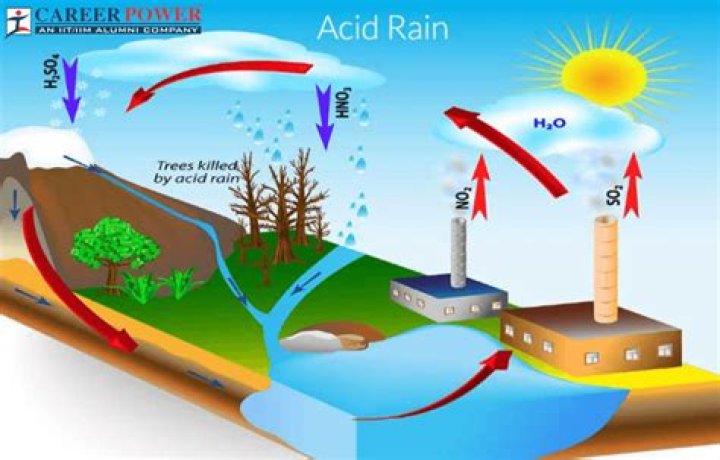
Acid rain results when sulfur dioxide (SO2) and nitrogen oxides (NOX) are emitted into the atmosphere and transported by wind and air currents. The SO2 and NOX react with water, oxygen and other chemicals to form sulfuric and nitric acids. These then mix with water and other materials before falling to the ground.
What are the main gas contributors to acid rain?
Power plants release the majority of sulfur dioxide and much of the nitrogen oxides when they burn fossil fuels, such as coal, to produce electricity. In addition, the exhaust from cars, trucks, and buses releases nitrogen oxides and sulfur dioxide into the air. These pollutants cause acid rain.
Does gas contribute to acid rain pollution?
But nitrogen oxides contribute to the formation of ground-level ozone, a major pollutant that can be harmful to people. Both gases cause environmental and health concerns because they can spread easily via air pollution and acid rain. … (Learn more about the effects of water pollution.)
Which pollutant contributes the most to acid rain?
Sulfur dioxide (SO2) and nitrogen oxides (NOx) released into the air by fossil-fuel power plants, vehicles and oil refineries are the biggest cause of acid rain today, according to the EPA. Two thirds of sulfur dioxide and one fourth of nitrogen oxide found in the atmosphere come from electric power generators.Is co2 responsible for acid rain?
Because carbonic acid is a relatively weak acid, the ability of carbon dioxide alone to generate true “acid rain” is very limited. Acid rain is caused by industrial emissions of sulfur dioxide and nitrogen oxides (which form much stronger acids when equilibrated in rainwater).
What are primary pollutants and secondary pollutants?
A primary pollutant is an air pollutant emitted directly from a source. A secondary pollutant is not directly emitted as such, but forms when other pollutants (primary pollutants) react in the atmosphere.
Which of the following gas is not responsible for acid rain?
Carbon dioxide reacts with water to form carbonic acid thus contributing to acid rain. So, from all these gases only carbon monoxide does not react with water to form an acid. Thus, not contributing to the acid rain, So, the correct answer is “Option C”.
Is CO2 primary pollutant?
Carbon Monoxide (CO) Carbon monoxide is released from volcanoes and forest fires as well. Secondary pollutants like ozone and carbon dioxide (CO2), a greenhouse gas, come from carbon monoxide.Is acid rain a primary pollutant?
Primary pollutants include ammonia, sulfur dioxide, nitrogen dioxide and carbon monoxide. Secondary pollutants include ground-level ozone, acid rain and nutrient enrichment compounds.
What are 3 effects of acid rain?It has been shown that acid rain has detrimental effects on trees, freshwaters and soils, destroys insects and aquatic life-forms, causes paint to peel, corrosion of steel structures such as bridges, and weathering of stone buildings and sculptures, as well as impacts on human health.
Article first time published onWhat pH does acid rain have?
However, when rain combines with sulfur dioxide or nitrogen oxides—produced from power plants and automobiles—the rain becomes much more acidic. Typical acid rain has a pH value of 4.0. A decrease in pH values from 5.0 to 4.0 means that the acidity is 10 times greater.
Which pollutant gas binds to Haemoglobin and prevents it from carrying oxygen?
Carbon monoxide as a pollutant Carbon monoxide is a poisonous gas which binds to haemoglobin in red blood cells, preventing them from carrying oxygen to the cells in the body.
Which gas is responsible for global warming?
Global Warming Potential (100-year): 1 Carbon dioxide (CO2) is the primary greenhouse gas emitted through human activities. In 2019, CO2 accounted for about 80 percent of all U.S. greenhouse gas emissions from human activities.
Which gas contributes the most to greenhouse effect?
In the effort to understand and address global climate change, most analysis has focused on rapidly rising emissions of carbon dioxide (CO2) and options for reducing them. Indeed, carbon dioxide, a byproduct of fossil fuel combustion, is the principal greenhouse gas contributing to global warming.
How do greenhouse gases cause acid rain?
According to the Environmental Protection Agency (EPA), acid rain is created when sulfur dioxide (SO2) and nitrogen oxides (NOx), as well as particulate matter (very fine particles), undergo a chemical reaction when exposed to water and oxygen. This reaction creates airborne sulfuric and nitric acids – aka, acid rain.
Which of the following pollutant causes acid rain Mcq?
Air Pollution MCQ Question 7 Detailed Solution The correct answer is Sulphur and Nitrogen. Acid rain is caused by oxides of Sulphur and Nitrogen. Acid rain results when Sulphur dioxide (SO2) and Nitrogen Oxides (NOx) are emitted into the atmosphere and transported by wind and air currents.
Does methane contribute to acid rain?
These non-toxic gases, most of which occur naturally in the atmosphere and are not pollutants, include carbon dioxide, methane, CFCs, and nitrous oxide. Acid rain is caused by toxic industrial smoke containing sulphur dioxide, nitrogen oxides, and chloride.
Does ammonia contribute to acid rain?
Before it returns to the ground in rain, ammonia gas in the atmosphere reacts with sulfuric or nitric acid vapors to form small particles known as aerosols, which are largely composed of ammonia sulfate. … At the moment, the Environmental Protection Agency does not recognize ammonia as an air pollutant.
What are the 5 main pollutants?
- Ozone (O3)
- Nitrogen Oxides (NOx)
- Carbon Monoxide (CO)
- Sulfur Dioxide (SO2)
- Particulate Matter (PM10 and PM2.5)
Which of the following pollutants are considered primary pollutants?
The five primary air pollutants include carbon monoxide, nitrogen oxides, particulate matter, sulfur oxides, and volatile organic compounds. The sources for all five of these pollutants include electricity production, industry, and transportation.
Which of the following pollutants is the major contributor to photochemical smog?
7. Which of the following pollutants is the major contributor to photochemical smog? Explanation: Ozone is the major constituent of photo-chemical smog.
Which of the following agent is mainly responsible for the secondary pollutants?
3. Which of the following is called the secondary air pollutant? Answer: (b) Ozone.
Which of the following is secondary pollutants?
Secondary pollutants include ozone, which is formed when hydrocarbons (HC) and nitrogen oxides (NOx) combine in the presence of sunlight. NO2, which is formed when NO combines with oxygen in the air. Acid rain, which is formed when sulfur dioxide or nitrogen oxides react with water, sulfuric acid etc.
Which are the major air pollutants?
Major air pollutants include carbon monoxide (CO), ammonia (NH3), nitric oxide (NO), nitrogen dioxide (NO2), ozone (O3), particulate matter (PM), sulphur dioxide (SO2) and volatile organic compounds (VOC).
What type of pollutant is carbon dioxide?
In its decision, the EPA stressed that it considers CO2 and other so-called greenhouse gases to be pollutants because of their role in propagating climate change, not because of any direct health effects.
Is methane a secondary pollutant?
The major primary pollutants are Oxides of Sulphur, Oxides of Nitrogen, Oxides of Carbon, Particulate Matter, Methane, Ammonia, Chlorofluorocarbons, Toxic metals etc. The secondary pollutants are not emitted directly.
What are some environmental impacts of acid rain?
The ecological effects of acid rain are most clearly seen in aquatic environments, such as streams, lakes, and marshes where it can be harmful to fish and other wildlife. As it flows through the soil, acidic rain water can leach aluminum from soil clay particles and then flow into streams and lakes.
What are the economic impacts of acid rain?
Power generating plants and other major producers of acid rain discharge their oxide wastes into the air in order to reduce production costs. This cost saving enables the producer to increase profits and expand production at the expense of those who suffer from acid rain.
Do volcanic eruptions cause acid rain?
Volcanic gases that pose the greatest potential hazards are sulfur dioxide, carbon dioxide, and hydrogen fluoride. Locally, sulfur dioxide gas can lead to acid rain and air pollution downwind from a volcano. These gases can come from lava flows as well as a volcano that erupts violently.
What is the major source of sulfur dioxide emissions?
Most of the sulfur dioxide released into the environment comes from electric utilities, especially those that burn coal. Some other sources of sulfur dioxide include petroleum refineries, cement manufacturing, paper pulp manufacturing, and metal smelting and processing facilities.
What pollutant gas causes respiratory problems?
Nitrogen dioxide is toxic. It can cause bronchitis and other lung diseases. It also dissolves in water in the clouds, forming an acidic solution that contributes to acid rain.