A strong base is something like sodium hydroxide or potassium hydroxide which is fully ionic. You can think of the compound as being 100% split up into metal ions and hydroxide ions in solution. Each mole of sodium hydroxide dissolves to give a mole of hydroxide ions in solution.
Are hydroxides weak bases?
Aqueous solutions of all Group 1 hydroxides are strong bases. Sodium is a group 1 metal. Sodium hydroxide is a strong base. Ammonia in aqueous solution is a weak base.
Is hydroxide a hard base?
Common examples of strong Arrhenius bases are the hydroxides of alkali metals and alkaline earth metals such as NaOH and Ca(OH)2. Strong bases are capable of deprotonating weak acids; very strong bases can deprotonate very weakly acidic C–H groups in the absence of water.
Is hydroxide a strong or weak acid?
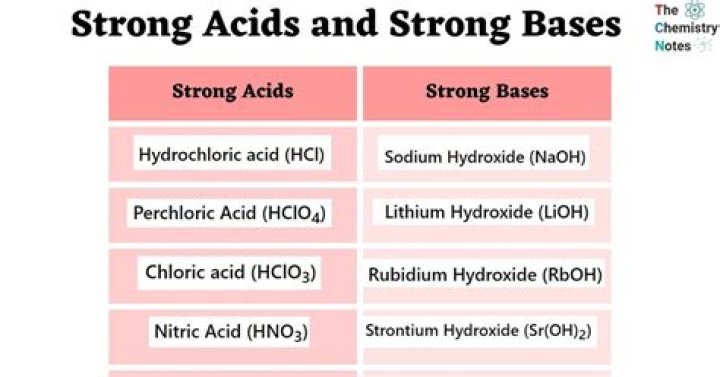
Similarly, there are only a few strong bases: lithium hydroxide (LiOH), sodium hydroxide (NaOH), potassium hydroxide (KOH), calcium hydroxide (Ca[OH]2), strontium hydroxide (Sr[OH]2), and barium hydroxide (Ba[OH]2). These strong acids and bases are 100% ionized in aqueous solution.Is OH 1 a strong base?
OH– is a base because of its “proton accepting” ability in aqueous solution, also, when OH– ions are dissolved in solution, the pH value of the solution gradually rises which shows its basic nature. The conjugate acid of OH– is H2O as conjugate acid is formed when H+ is added to the given species.
Why is NaOH a strong base?
Strong bases are characterized by the fact that they dissociate completely in aqueous solution. In this case, sodium hydroxide, NaOH , is classified as a strong base because it dissociates completely in aqueous solution to form sodium cations, Na+ , and hydroxide anions, OH− .
Why is caoh2 a strong base?
Ca(OH)2 is the strong base because it completely dissociates in an aqueous solution to yield OH– ion and no moles of it remain undissociated inside the solution. And the amount of OH– ions in an aqueous solution is very high and we know OH– ions have a tendency to accept the proton.
What makes a strong base?
A strong base is a base that is completely dissociated in an aqueous solution. These compounds ionize in water to yield one or more hydroxide ion (OH-) per molecule of base. … Strong bases react with strong acids to form stable compounds.Is CsOH a strong base?
AcidsBasesH2SO4CsOHHClO3Mg(OH)2HClO4Ca(OH)2Sr(OH)2
Is MGOH a base?The compound Mg(OH)2 (magnesium hydroxide) is a weak base.
Article first time published onWhich is not a strong base?
Zn(OH)2 is not a strong base among the following.
What is strong base example?
- Potassium hydroxide (KOH)
- Sodium hydroxide (NaOH)
- Barium hydroxide (Ba(OH)2)
- Caesium hydroxide (CsOH)
- Sodium hydroxide (NaOH)
- Strontium hydroxide (Sr(OH)2)
- Calcium hydroxide (Ca(OH)2)
- Lithium hydroxide (LiOH)
What substances are bases?
Examples of bases are the hydroxides of the alkali and alkaline earth metals (sodium, calcium, etc.) and the water solutions of ammonia or its organic derivatives (amines). Such substances produce hydroxide ions (OH-) in water solutions (see Arrhenius theory).
Is caoh2 a strong base?
Calcium hydroxide still counts as a strong base because of that 100% ionisation. In the presence of extra hydroxide ions from, say, sodium hydroxide, the equilibrium is still there, but the position of equilibrium has been shifted well to the left according to Le Chatelier’s Principle.
Is caoh2 a strong base or a weak base?
Calcium hydroxide is a strong base. Since it produces OH- ions on electrolyte dissociation.
Is caoh2 a strong alkali?
Complete answer: Calcium hydroxide is a strong base, however less soluble in water. It gets ionized 100% into calcium ions and hydroxide ions. value is 1.37 (First OH−) and 2.43 (second OH−). … Therefore, the concentration of hydroxide ions in a strongly basic solution is equal to that of the undissociated base.
Is NaOH a strong bond?
Sodium hydroxide (NaOH) is strong base because it fully dissociates in water to produce hydroxide ions.
Is NaOH a strong base or acid?
NaOH is a strong base.
Which is strong base NaOH or CsOH?
NaOH and CsOH are both fully ionised hydroxides so there is no difference. For a stronger base something stronger than the hydroxide is needed such as an alkoxide ion. Sodium ethoxide (Na OC2H5), for example, is a stronger base than sodium hydroxide.
Is RbOH strong base?
Strong bases are capable of deprotonating weak acids; very strong bases can deprotonate very weakly acidic C–H groups in the absence of water. RbOH is Rubidium hydroxide, it is a strong base.
Which is stronger base LIOH or NaOH?
NaOH is more basic than LIOH.
Why is ammonium hydroxide a weak base?
Ammonium hydroxide is a weak base because it does not dissociate completely into ions.
What's the strongest base?
The title of strongest base in the world belongs to ortho-diethynylbenzene dianion. This superbase has the strongest proton affinity ever calculated (1843 kJ mol−1), beating out a long-standing contender known as lithium monoxide anion. Watch the video to learn more about bases and superbases!
Which metal hydroxide is the strongest base?
The hydroxides of the alkali metals, lithium, sodium, potassium, rubidium, and cesium, are the strongest bases and the most stable and most soluble of the hydroxides. Sodium hydroxide, NaOH, also known as caustic soda or lye, is of great industrial importance.
Is zinc hydroxide a strong base?
Zn(OH)2Zinc hydroxideDensity3.05 g/cm³Molecular Weight/ Molar Mass99.424 g/molMelting Point125° C (257° F)Chemical FormulaH2O2Zn
Is magnesium hydroxide a strong base than calcium hydroxide?
See Calcium hydroxide (Ca(OH)2) is more basic than magnesium hydroxide (Mg(OH)2). Since the size of calcium is more than magnesium so it is easy of Calcium to remove hydroxide group, since the bond strength is weaker than that in case of Mg(OH)2. Hence it is more basic.
Is CH3CH2NH2 a strong base?
CH3CH2NH2 is a weak base, and HCl is a strong acid.
Is sodium hydroxide a base?
General information. Sodium hydroxide (caustic soda) is highly soluble in water, and sodium hydroxide solutions are strong bases.
Which of the following is a strong base NaOH?
Sodium hydroxide is a strong base because it dissociates completely in an aqueous solution to form sodium cations, Na+, and hydroxide anions, OH−. Sodium hydroxide is completely ionic, containing sodium ions and hydroxide ions.
Is sodium hydroxide a weak base?
Because sodium hydroxide is a strong base that dissociates completely in solution to form hydroxide ions, if the concentration of hydroxide ions in solution is .
Are all metal hydroxides strong bases?
Metal hydroxides are hydroxides of metals. They are often strong bases. … Some metal hydroxides, such as alkali metal hydroxides, ionize completely when dissolved. Certain metal hydroxides are weak electrolytes and dissolve only partially in aqueous solution.