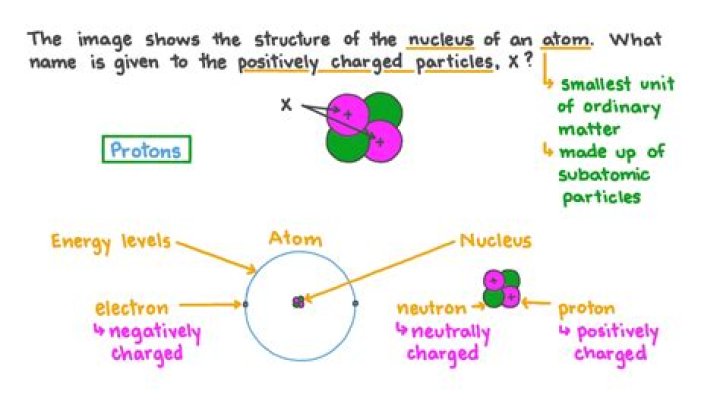
Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they’re neutral).
What is the positively charge particle in an atom called?
Protons are positively charged particles found within atomic nuclei. Rutherford discovered them in experiments with cathode-ray tubes that were conducted between 1911 and 1919. Protons are about 99.86% as massive as neutrons according to the Jefferson Lab. The number of protons in an atom is unique to each element.
What is the negatively charged particle of an atom?
Subatomic particles include electrons, the negatively charged, almost massless particles that nevertheless account for most of the size of the atom, and they include the heavier building blocks of the small but very dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.
Is an ion a positively charged particle?
By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. When an ion is formed, the number of protons does not change.How do you know if an atom is positive or negative?
If the atom has more electrons than protons, it is a negative ion, or ANION. If it has more protons than electrons,it is a positive ion.
How do you know if an ion is positively charged?
The positive charge (more protons versus electrons) for a cation is shown by a number and plus sign after the formula. If there’s just a plus sign, it means the charge is plus 1.
What's a positively charged ion?
A cation is an ion with positive charge, which means it has more protons (positively-charged particles) than electrons (negatively-charged particles). Cations are formed when an atom loses one or more electrons: the loss of the negatively-charged electron(s) results in an overall positive charge.
What makes a particle positive or negative?
The most common charged particles are the electron and proton. Atoms with an excess of electrons are called negative ions. Those with missing electrons are called positive ions.Are particles of an atom that are electrically neutral?
An atom is electrically neutral because the overall charge of an atom is zero. The atoms are made of three subatomic particles called protons, electrons and neutrons. … The charge of both protons and electrons are of equal strength, therefore atoms having an equal number of protons and electrons are electrically neutral.
What has a neutral charge in an atom?A neutral charge is the electrochemical occurrence where an atom has an equal number of electrons and protons. Such an atom is neither positively charged nor negatively charged because the protons and electrons balance each other out.
Article first time published onWhich particle is a negatively charged ion?
Electron: A negatively charged particle found circling or orbiting an atomic nucleus. An electron, like a proton is a charged particle, although opposite in sign, but unlike a proton, an electron has negligible atomic mass.
What are negative and positive ions?
Ions are invisible charged particles in the air – either molecules or atoms, which bear an electric charge. … To put it simply, positive ions are molecules that have lost one or more electrons whereas negative ions are actually oxygen atoms with extra-negatively-charged electrons.
How is a positively charged ion formed?
Ions form when atoms gain or lose electrons. Since electrons are negatively charged, an atom that loses one or more electrons will become positively charged; an atom that gains one or more electrons becomes negatively charged.
Which atom will form singly positively charged ion?
#For H: EC: 2(K),8(L),1(M). it can readily lose one electron to attain stable inert gas configuration. hence it forms singly positively charged ion.
Why the nucleus of an atom is positively charged?
The nucleus has an overall positive charge as it contains the protons. Every atom has no overall charge (neutral). This is because they contain equal numbers of positive protons and negative electrons. These opposite charges cancel each other out making the atom neutral.
Why is an atom considered to be neutral?
When an atom has an equal number of electrons and protons, it has an equal number of negative electric charges (the electrons) and positive electric charges (the protons). The total electric charge of the atom is therefore zero and the atom is said to be neutral.
What gives an electron its negative charge?
I can’t really answer this question, but based on experiments carried out by scientists, electrons have a negative charge because they are attracted to anodes (a positively charged electrode). We know that opposite charges attract. Electrons are known as fundamental particles. Electron does not acquire charge.
Which one is positively charged particle?
A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.
What is negative particle?
In English grammar, the negative particle is the word not (or its reduced form, -n’t) used to indicate negation, denial, refusal, or prohibition. Also called a negative adverb. The most usual way in which negative sentences are constructed in English is with the negative particle not or n’t.
What gives a particle charge?
Electric charge is carried by subatomic particles. In ordinary matter, negative charge is carried by electrons, and positive charge is carried by the protons in the nuclei of atoms. … The proton has a charge of +e, and the electron has a charge of −e.
How can an atom become positively charged?
When one or more electrons is stripped away from an atom, it becomes positively charged. Some atoms can attract additional electrons so they become negatively charged. Atoms which are not electrically neutral are called ions. One can collect electric charge by transferring electrons.
Where are positive ions?
In nature, positive ions are commonly formed by strong winds, dust, humidity and pollution. They are at their highest levels just before an electrical storm.
Do electronics give off positive ions?
What Generates Positive Ions? In general, anything that’s toxic or has electromagnetic capabilities will generate harmful positive ions including: Electronic devices like televisions, phones, and computers. Fluorescent Lighting.