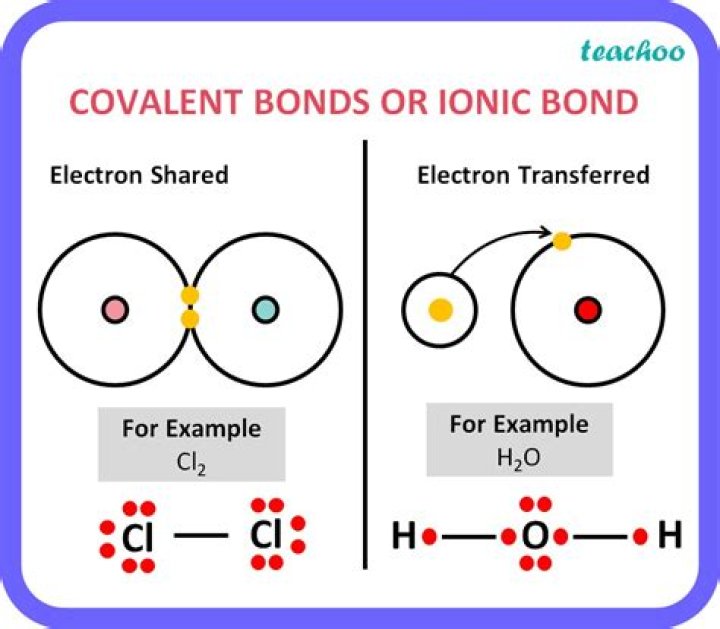
If a compound is made from a metal and a non-metal, its bonding will be ionic.If a compound is made from two non-metals, its bonding will be covalent.
How do you tell if a compound is both ionic and covalent?
A dead giveaway the compound contains both bonds is when it has a metal cation bonded to an anion that only contains nonmetals. Also, any compound that contains the ammonium (NH4+) cation has both ionic and covalent bonds. The nitrogen and hydrogen atoms are joined by covalent bonds.
What is the easiest way to tell the difference between an ionic and a covalent bond?
By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.
What compounds have both ionic and covalent bonding?
Calcium carbonate is another example of a compound with both ionic and covalent bonds. Here calcium acts as the cation, with the carbonate species as the anion. These species share an ionic bond, while the carbon and oxygen atoms in carbonate are covalently bonded.How do you know if a compound is ionic or covalent when looking at a chemical formula like NaCl?
Form the molecular formula, you can compare the Electronegativity (EN) between the atoms. If the Electronegativity Difference (∆EN)>1.8, then the compound will be ionic. But if the ∆EN<1.8,then the compound might be covalent. In NaCl, the electronegativity of sodium & chlorine are 0.93 & 3.16 respectively.
Is CH4 ionic or covalent?
Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram). The lines, or sticks, as we say, represent the covalent bonds. There are four bonds from a central carbon (C) linking or bonding it to four hydrogen atoms (H).
Does nacl contain both ionic and covalent bonds?
PropertyIonicCovalentMelting temperatureHighLow
Which of the following compounds contains both ionic and covalent bonds AP Chem?
Therefore, the compound that contains both ionic and covalent bonds is (D) LiOH.Can a compound have both ionic and covalent bonds explain your answer?
A compound can have both ionic and covalent bonds if the covalent bonds are part of a polyatomic ion.
Which of the following compound contains both covalent and co ordinate bond?C2H5NC.
Article first time published onHow do you know if a compound is ionic?
- If a compound is made from a metal and a non-metal, its bonding will be ionic.
- If a compound is made from two non-metals, its bonding will be covalent.
How are molecules different from ionic compounds?
Molecular compounds are pure substances formed when atoms are linked together by sharing of electrons while ionic compounds are formed due to the transfer of electrons. … Molecular compounds are made due to covalent bonding while ionic compounds are made due to ionic bonding.
How do you predict ionic compounds?
Determining Ionic Charge. The charge that will be on an ion can be predicted for most of the monatomic ions. Many of these ionic charges can be predicted for entire families of elements.
Which two compounds are molecules which both contain a double bond?
Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist between two different elements: for example, in a carbonyl group between a carbon atom and an oxygen atom. Other common double bonds are found in azo compounds (N=N), imines (C=N), and sulfoxides (S=O).
Does CH4 contain both ionic and covalent bonds?
CH4 or methane is a purely covalent compound or a non ionic compound. As the name suggests , it has no ions.it is formed by the sharing of electrons between each H atom and 1 C atom thus forming 4 covalent CH bonds.
Does NaOH have both ionic and covalent bonds?
Bonding in NaOH. Both ionic and covalent bond is present in sodium hydroxide. The bond between Na and O is ionic the bond between O and H is covalent.
Is cacl2 an ionic or covalent bond?
Calcium chloride is an ionic compound.
Does CaO have an ionic bond?
The ionic formula for calcium oxide is simply CaO. Oxygen has six valence electrons and is looking to gain two electrons to complete the octet (8) electron count in the valence shell making it a -2 anion.
Is O2 a compound?
The oxygen molecule O2 is considered a molecule but not a compound. This is because O2 is made of two atoms…
Are molecules ionic or covalent?
Compounds can be classified as ionic or covalent. Molecules are the simplest unit of a covalent compound, and molecules can be represented in many different ways. Atoms are the smallest units of matter that still retain the fundamental chemical properties of an element.
Which pure substance below contains both covalent and ionic bonding?
CaSO4: Calcium sulfate (or calcium sulphate). Also remember also polyatomic ions (relating to a molecule made up of more than two atoms.) are held together by covalent bonds, so this compound contains both ionic and covalent bonds.
Does co2 contain both ionic and covalent bonds?
Note that carbon dioxide has two covalent bonds between each oxygen atom and the carbon atom, which is shown here as two lines and referred to as a double bond. When molecules are symmetrical, however, the atoms pull equally on the electrons and the charge distribution is uniform. Symmetrical molecules are nonpolar.
Does MgCl2 have both ionic and covalent bonds?
MgCl2 is an ionic compound.
Is NH4Cl ionic or covalent?
Thus, NH4Cl and (NH4)2SO4 (both solids) are ionic compounds.
Which of the following claims about a binary compound in which bonding is ionic is most likely to be scientifically valid?
Which of the following claims about a binary compound in which the bonding is ionic is most likely to be scientifically valid? There is equal sharing of electrons between the atoms of the elements in the compound.
What is the difference between covalent bonds and ionic bonds?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions.
Does H2O contain both covalent and coordinate bond?
There is no dative bond in H2O, oxygen is attached to two hydrogen atoms through two covalent bonds and there are two lone electron pairs on oxygen atom.
Which of the following pairs contains only covalent bond?
Examples of compounds that contain only covalent bonds are methane (CH4), carbon monoxide (CO), and iodine monobromide (IBr). Covalent bonding between hydrogen atoms: Since each hydrogen atom has one electron, they are able to fill their outermost shells by sharing a pair of electrons through a covalent bond.
How do you identify a covalent compound?
If the difference in electronegativities between two bonding elements is less than 0.5, then you have a nonpolar covalent bond. If the difference in electronegativities between two bonding elements is between 0.5 and 1.6, then you have a polar covalent bond.
Are covalent and molecular compounds the same?
Molecular compounds or covalent compounds are those in which the elements share electrons via covalent bonds. The only type of molecular compound a chemistry student is expected to be able to name is a binary covalent compound. This is a covalent compound made up of only two different elements.
How do you determine the type of bond in a compound?
- Look at the chemical formula.
- Identify the elements in the compound.
- Determine if the elements are metals or nonmetals (using a periodic table)
- Metal – Metal = Metallic.
- Metal – Nonmetal = Ionic.
- Nonmetal — Nonmetal = Covalent.