Grignard reagents add to carbonyl compounds to give primary, secondary, and tertiary alcohols. A primary alcohol is synthesized by reacting the Grignard reagent, R′─MgX, with formaldehyde. Reacting a Grignard reagent with an aldehyde gives a secondary alcohol.
How does Grignard reagent break?
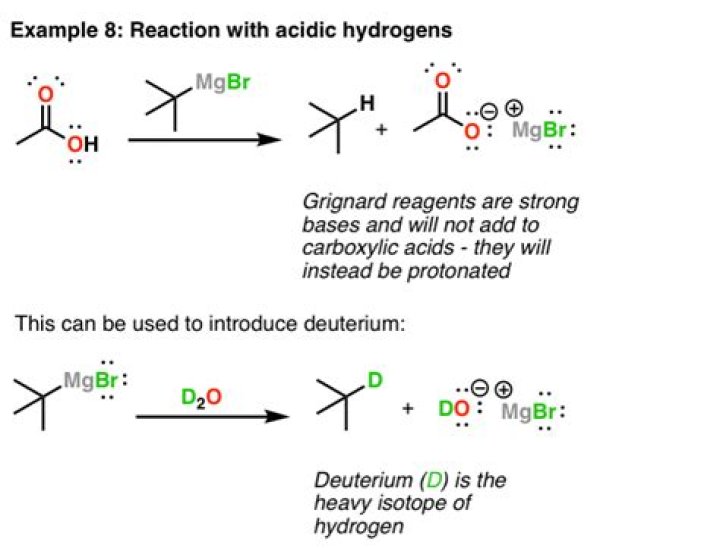
Grignards are destroyed in the presence of carboxylic acids. While we can react them with derivatives, carboxylic acids are too acidic and will destroy the Grignard, just as the Grignard would attack any acid or polar protic solvent.
What are Grignard reagents give a chemical reaction for their preparation?
Grignard reagents are formed by the reaction of magnesium metal with alkyl or alkyl halides. They are wonderful nucleophiles, reacting with electrophiles such as carbonyl compounds (aldehydes, ketones, esters, carbon dioxide, etc.) and epoxides.
What does a Grignard reagent do to an ester?
An ester (or an acid halide or an anhydride) reacts first with a Grignard reagent to form a ketone, which reacts further to give an alcohol. It is not usually possible to make and isolate a ketone through reaction of an ester or other acid derivative with only one mole of Grignard reagent.Are Grignard reagents strong bases?
As discussed above, Grignard and organolithium reagents are powerful bases. Because of this they cannot be used as nucleophiles on compounds which contain acidic hydrogens. If they are used they will act as a base and deprotonate the acidic hydrogen rather than act as a nucleophile and attack the carbonyl.
How do you test a Grignard reagent?
To confirm Grignard Reagent before step up to next reaction add Iodine solution . The decolorization of iodine indicates the reagent has been formed.
Why are organometallic reagents destroyed in water?
Water or alcohols would protonate and thus destroy the Grignard reagent, because the Grignard carbon is highly nucleophilic. This would form a hydrocarbon.
How do you store Grignard reagents?
Grignard reagents aren’t very stable and should be used as soon as possible. Short term storage can be done in a flask filled with dry argon, kept in a cold place, like a fridge.How do you initiate Grignard?
Usually you take magnesium under nitrogen and you heat it for 10-15 minutes (usually with an heat gun). Then in dry THF you add as activator few mg of iodine or few microliter of 1,2-dibromoethane. I used both method and I’ve never had any problem in the generation of the Grignard reagent.
Does Grignard reagent react with alcohol?Grignard reagents react rapidly with acidic hydrogen atoms in molecules such as alcohols and water to produce alkanes.
Article first time published onWhat functional groups react with Grignard reagents?
Grignard reagents are formed by the reaction of magnesium metal with alkyl or alkenyl halides. They’re extremely good nucleophiles, reacting with electrophiles such as carbonyl compounds (aldehydes, ketones, esters, carbon dioxide, etc) and epoxides.
Do Grignards react with alkenes?
This modification requires aryl or vinyl Grignards. Adding just the Grignard and the alkene does not result in a reaction demonstrating that the presence of oxygen is essential.
How is isopropyl alcohol prepared from Grignard reagent?
Ethanal is a compound which has two carbon atoms and the product isopropyl alcohol has three carbon atoms. This is made by reacting ethanal with Grignard reagent. The chemical formula of Grignard reagent is RMgBr, where R is methyl group. … The intermediate undergoes hydrolysis to give isopropyl alcohol.
Which of the following reagents will give a primary alcohol on reacting with a Grignard reagent?
The reaction formaldehyde with Grignard reagent gives primary alcohol.
What do Grignard reagent not react with?
Grignard reagent generally doesn’t reacts with alkene due to non occurrence of acidic hydrogen in alkenes. Acidic hydrogen are those which are connected to highly electronegative element or to sp hybridised carbon as in alkynes.
Why is magnesium used in Grignard reagent?
In addition, evidence suggests that the ether molecules actually coordinate with and help stabilize the Grignard reagent: The magnesium metal used in the synthesis contains a layer of oxide on the surface that prevents it from reacting with the alkyl bromide.
Which reagent is the strongest base?
- LiOH – lithium hydroxide.
- NaOH – sodium hydroxide.
- KOH – potassium hydroxide.
- RbOH – rubidium hydroxide.
- CsOH – cesium hydroxide.
- *Ca(OH)2 – calcium hydroxide.
- *Sr(OH)2 – strontium hydroxide.
- *Ba(OH)2 – barium hydroxide.
Which of the following are organometallic reagents?
Organometallic reagents such as phenylmagnesium bromide and methyl lithium are among the strongest bases there are. Consequently they will deprotonate compounds such as amines, alcohols, and carboxylic acids. Figure 2 presents one reaction that is representative of each of these situations.
Why is acid used to quench a Grignard reaction?
Grignard reagents are good nucleophiles and strong bases. Thus, they react with electrophiles and acids. … The addition of hydrochloric acid is necessary to quench the left over Grignard reagent and to convert the magnesium alcoholate into the alcohol.
What is Grignard reagent shaala?
Solution. Grignard’s reagent is an organometallic compound in which the divalent magnesium is directly linked to an alkyl group acid and a halogen atom. It is represented by general formula R–Mg–X. Concept: Hydrocarbons: Alkanes – Reactions of Haloarenes – Nucleophilic Substitution.
Which of the following is Grignard reagent?
Grignard reagent, any of numerous organic derivatives of magnesium (Mg) commonly represented by the general formula RMgX (in which R is a hydrocarbon radical: CH3, C2H5, C6H5, etc.; and X is a halogen atom, usually chlorine, bromine, or iodine).
How does iodine activated magnesium?
Various methods of magnesium surface activation are tested. For example, iodine and ferric chloride increase reactivity by increasing the density of reactive sites and by increasing the rate at which individual sites react.
What is the side reaction between Grignard reagent and Bromobenzene?
Reaction Scheme Biphenyl, which is formed from a coupling reaction between unreacted bromobenzene and Grignard reagent, is often observed as a major impurity in this reaction. Formation of the side product is favored by high concentrations of bromobenzene and increased reaction temperature.
How do you activate turning Mg?
Dry Stirring of the Magnesium Turnings. Some reports have appeared in the literature on activation by stirring the magnesium turnings in an inert atmosphere. During stirring the oxide layer on the magnesium turnings is reduced, leaving the activated metal surface.
Why do you add iodine to Grignard?
Addition of iodine is to help remove any MgO on the surface of the Mg. Removing MgO allows for Mg and the aryl/alkyl halide to come in contact and react. Sonication or addition of methyl iodide or 1,2-dibromoethane can also help with initiation.
Why do Grignard reactions have to be dry?
A typical Grignard reagent might be CH3CH2MgBr. … Everything must be perfectly dry because Grignard reagents react with water (see below). Any reactions using the Grignard reagent are carried out with the mixture produced from this reaction.
Why must Grignard reaction be anhydrous?
During the Grignard reaction, conditions must be anhydrous because the Grignard reagent reacts readily with water and forms alkanes. Grignard reagent must be kept away from water as it reacts readily with water, forming an alkane and MgBr .
How do you minimize the formation of biphenyl?
A slow addition also reduces the temperature in the reaction and suppresses the formation of biphenyl because the formation of the Grignard reagent is favored at lower temperatures. If he added the solution too rapidly, the reaction mixture would foam very heavily and turn dark brown.
How will you obtain Thio alcohol from Grignard reagent?
The Grignard Reaction is the addition of an organomagnesium halide (Grignard reagent) to a ketone or aldehyde, to form a tertiary or secondary alcohol, respectively. The reaction with formaldehyde leads to a primary alcohol.
How does a Grignard reagent act as a nucleophile?
Because organometallic reagents react as their corresponding carbanion, they are excellent nucleophiles. The basic reaction involves the nucleophilic attack of the carbanionic carbon in the organometallic reagent with the electrophilic carbon in the carbonyl to form alcohols.
What type of alcohol results when a Grignard reagent reacts with a ketone followed by h2o )?
Grignard reagents such as methylmagnesium bromide are therefore sources of a nucleophile that can attack the + end of the C=O. double bond in aldehydes and ketones. If we treat the product of this reaction with water, we get an tertiary alcohol.